
 is given by,
is given by,

 Knowledge
of specific heats and/or heat capacities and
the fact that energy must be conserved
allows us to determine the equilibrium
temperature of two objects initially at
different temperatures by demanding that,
Knowledge
of specific heats and/or heat capacities and
the fact that energy must be conserved
allows us to determine the equilibrium
temperature of two objects initially at
different temperatures by demanding that,
 Typically in a solid
the composite atoms or molecules are arranged in a rigid
lattice structure - each atom or molecule is largely fixed in
space.
Typically in a solid
the composite atoms or molecules are arranged in a rigid
lattice structure - each atom or molecule is largely fixed in
space.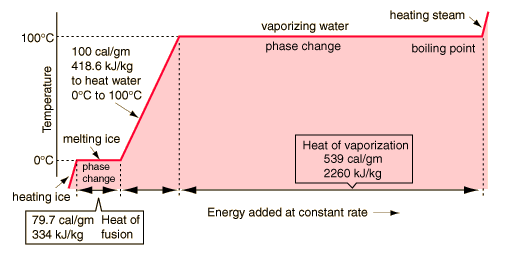


![]()
There is
this farmer who is having problems with his chickens. All of the
sudden, they are all getting very sick and he doesn't know what
is wrong with them. After trying all conventional means, he
calls a biologist, a chemist, and a physicist to see if they can
figure out what is wrong. So the biologist looks at the
chickens, examines them a bit, and says he has no clue what
could be wrong with them. Then the chemist takes some tests and
makes some measurements, but he can't come to any conclusions
either. So the physicist tries. He stands there and looks at the
chickens for a long time without touching them or anything. Then
all of the sudden he starts scribbling away in a notebook.
Finally, after several gruesome calculations, he exclaims, 'I've
got it! But it only works for spherical chickens in a vacuum.'
Dr. C. L. Davis
Physics Department
University of Louisville
email: c.l.davis@louisville.edu
