
 proposed a model of the Hydrogen atom which relied
primarily on classical ideas. This "Bohr
model" was successful in describing some of the basic properties of
the Hydrogen atom.
proposed a model of the Hydrogen atom which relied
primarily on classical ideas. This "Bohr
model" was successful in describing some of the basic properties of
the Hydrogen atom. The
Bohr model assumes that the Hydrogen atom electron moves in cirular
orbits about the proton, but that only orbits of particular
radii, rn are allowed where,
The
Bohr model assumes that the Hydrogen atom electron moves in cirular
orbits about the proton, but that only orbits of particular
radii, rn are allowed where,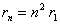
Dr. C. L. Davis
Physics Department
University of Louisville
email: c.l.davis@louisville.edu
